
(c) In the cathode ray, the beam (shown in yellow) comes from the. 
Previously, an atom was defined as the smallest part of an element that maintains the identity of that element. 4 ), is a fundamental concept that states that all elements are composed of atoms. He is known for the Thomson atomic theory. The modern atomic theory, proposed about 1803 by the English chemist John Dalton (Figure 1.5.4 1.5. 
Thomson is credited with the discovery of the electron, the negatively charged particle in the atom. Thomson is buried in Westminster Abbey, near Sir Isaac Newton. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. He died August 30, 1940, Cambridge, Cambridgeshire, England. 2.2: The Discovery of Atomic Structure is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. Thomson produced a visible beam in a cathode ray tube. Three important kinds of radiation are particles (helium nuclei), particles (electrons traveling at high speed), and rays (similar to x-rays but higher in energy). Chadwick was educated at the University of Manchester, where he worked under Ernest Rutherford and earned a master’s degree in 1913. 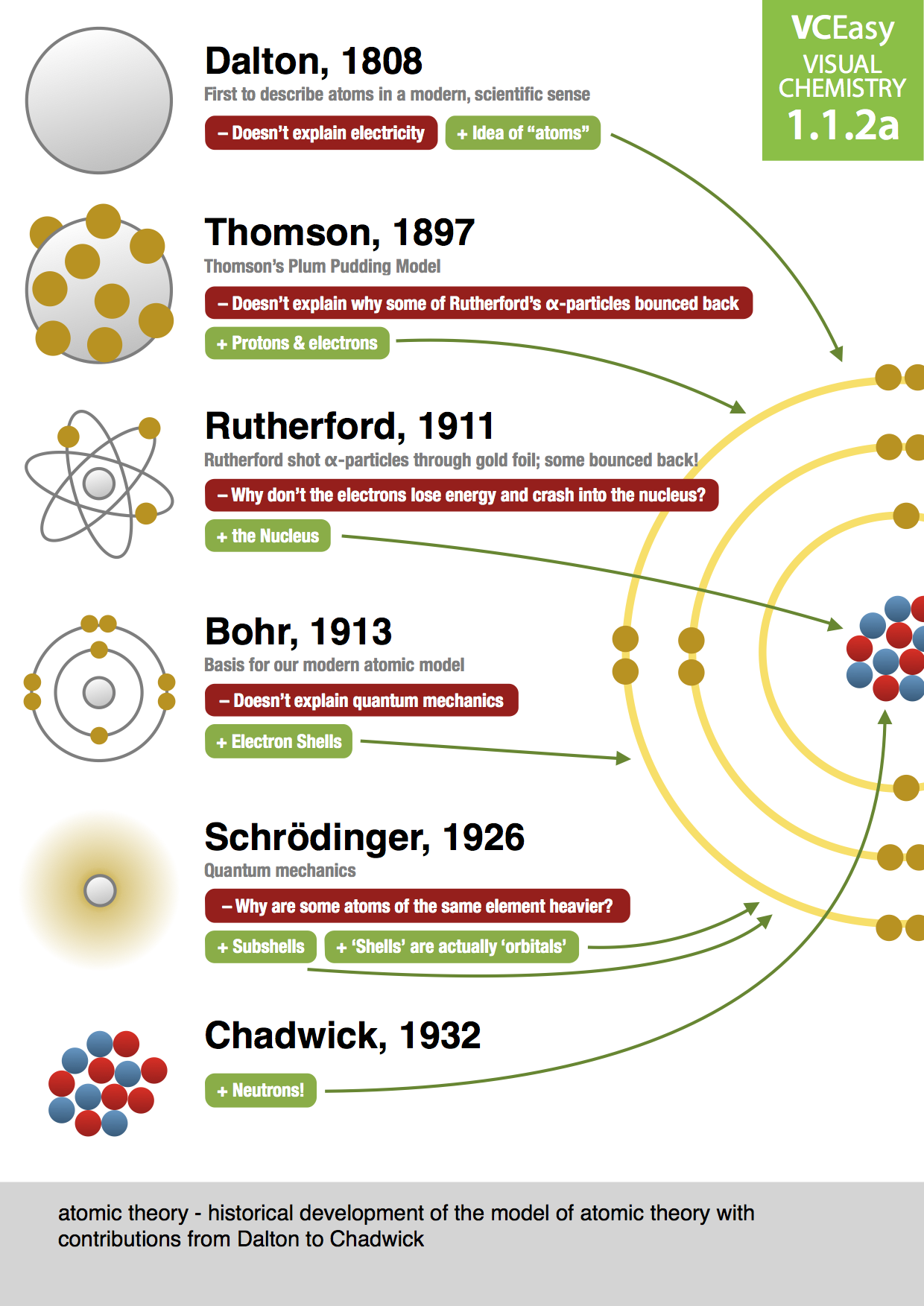
These scientists, among many others, have significantly contributed to our knowledge of the atomic theory and have played a vital role in the development of nuclear science. James Chadwick (born October 20, 1891, Manchester, Englanddied July 24, 1974, Cambridge, Cambridgeshire) English physicist who received the Nobel Prize for Physics in 1935 for the discovery of the neutron. (c) In the cathode ray, the beam (shown in yellow) comes from the cathode and is accelerated past the anode toward a fluorescent scale at the end of the tube. To account for the existence of isotopes, the second postulate of his atomic theory was modified to state that atoms of the same element must have identical chemical properties. James Chadwick discovered the neutron, an uncharged particle found in the nucleus of an atom, which revolutionized our understanding of atomic structure. (b) This is an early cathode ray tube, invented in 1897 by Ferdinand Braun. By the late 1800’s, John Dalton’s view of atoms as the smallest particles that made up all matter had held sway for about 100 years, but that idea was about to be challenged. Thomson produced a visible beam in a cathode ray tube. James Chadwick won a Nobel Prize in 1935 for his discovery of the neutron. The word 'atom' comes from the Greek word for uncuttable. If the spheres touch, they are part of a single unit of a compound. Atomic theory states that matter is composed of discrete units, called atoms. The purple spheres represent atoms of another element. Thomson produced a visible beam in a cathode ray tube. The results of these measurements indicated that these particles were much lighter than atoms (Figure 3.3.1 3.3. This module is an updated version of Atomic Theory I. In the following drawing, the green spheres represent atoms of a certain element. Trending Questions How is each triglyceride different from each others? How was Mt Schank formed? Did Mount Ararat ever erupted? How is a watershed related to a river system? What stimulated John McNeile Hunter to do the work he did? Date of battle of New Orleans? What is greater latitude? Why would a light switch have two black wires for power and two whites caped off? What causes protein to be denatured? When was the first thinning of the ozone layer? What part of the planet is the least explored? Which ones always fit together to make base pairs? What is the large country in central Africa borded by 9 countries? How can you bring your venus fly trap back to life? What the furthest point north on the Earth is called? Ideal conditions for photosynthesis? Which part of photosynthesis is water carbon dioxide and energy used? Your empty hand is not hurt when it bangs lightly against the wall.\): (a) J. In 1932, James Chadwick discovered the neutron a particle with no charge in the nucleus of the atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
